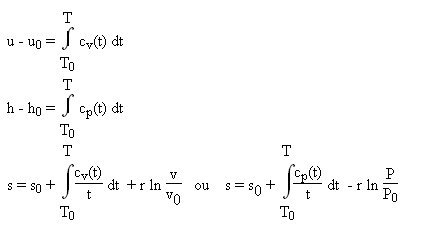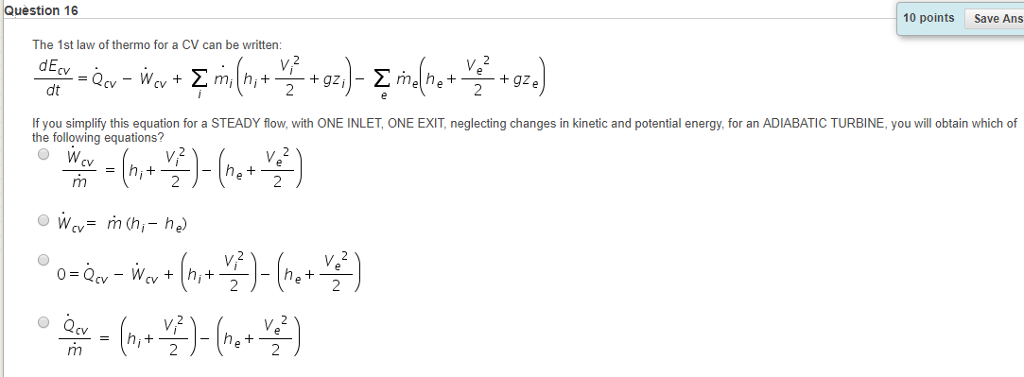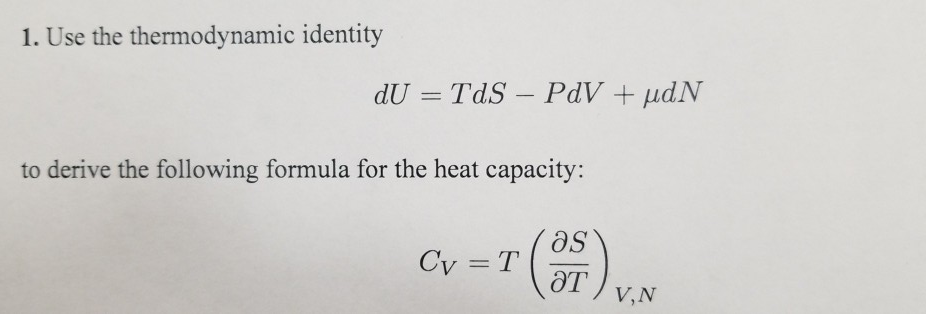Thermo Scientific™ 7000 Series Copolymer Microsphere Suspensions 222μm; CV ≤ 12%; 15mL Thermo Scientific™ 7000 Series Copolymer Microsphere Suspensions | Fisher Scientific
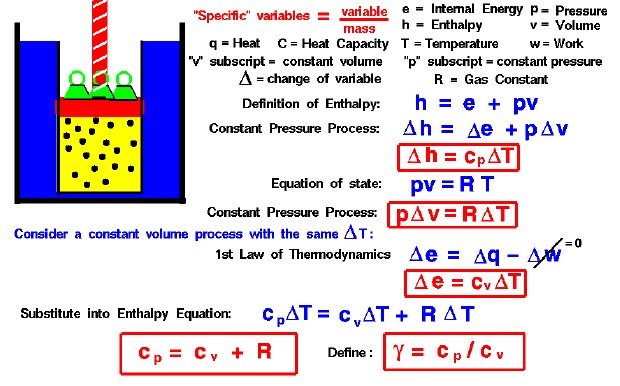
Use the thermodynamic relations to show that for an ideal gas CP − CV = R. - Sarthaks eConnect | Largest Online Education Community

SOLVED: 5. A general relation between Cp and Cy. (a) Using the thermodynamic identity, show that the heat capacity at a fixed volume V is expressed as: Cv = T (∂S/∂T)V (b)