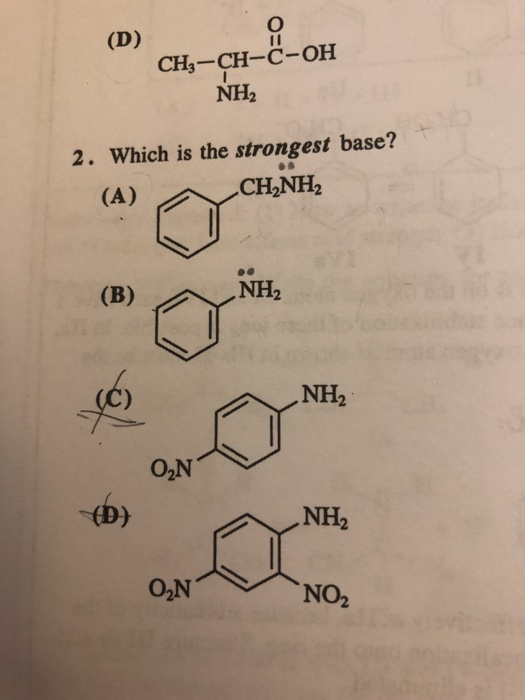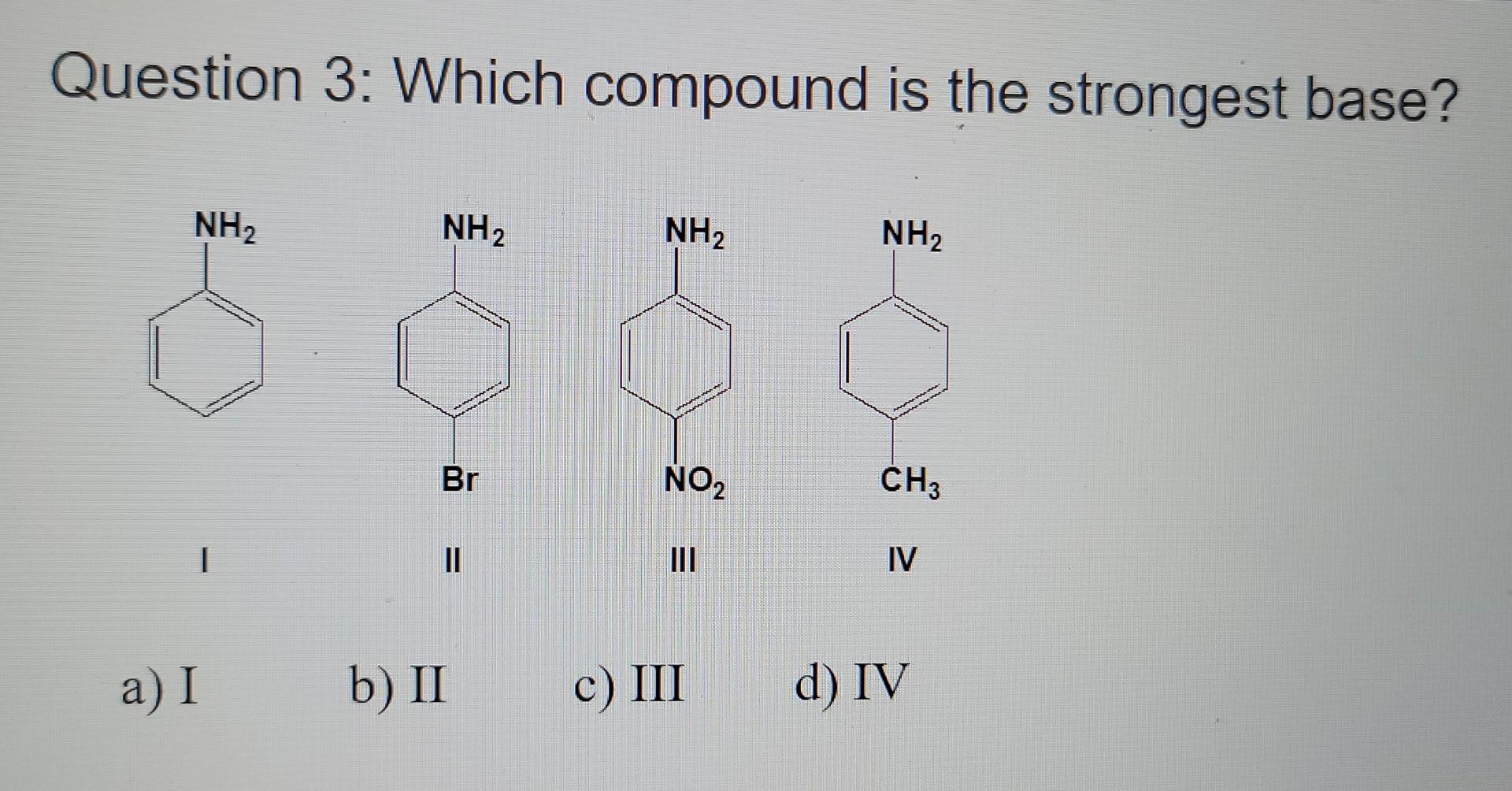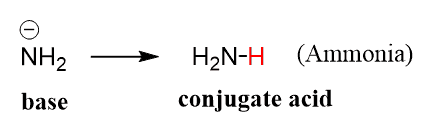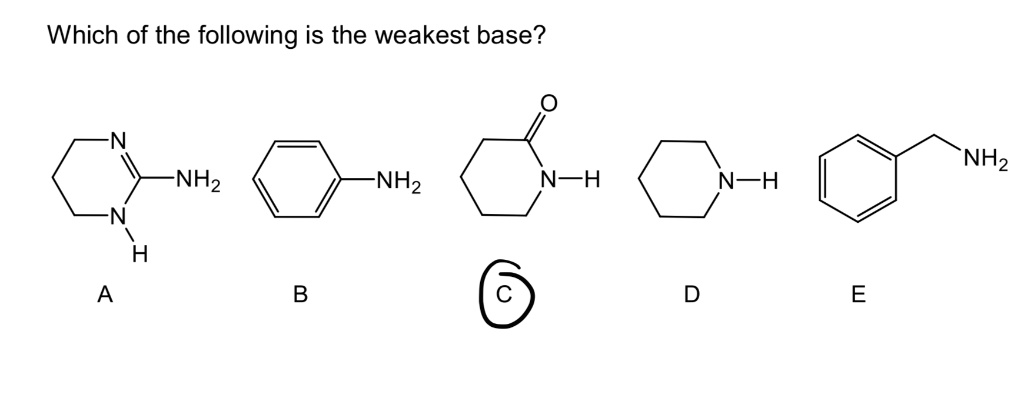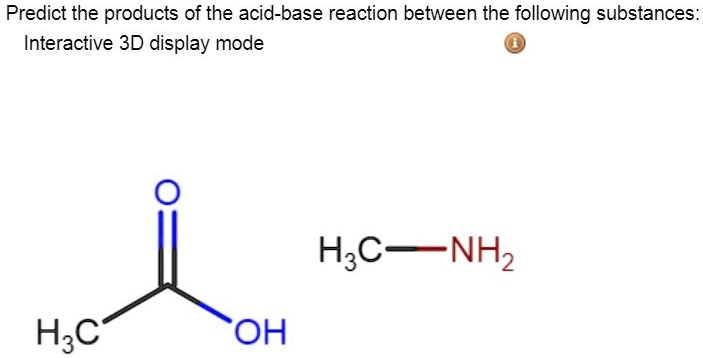
SOLVED: Predict the products of the acid-base reaction between the following substances: Interactive 3D display mode H3C NH2 H3C - OH

OneClass: Questions 3 and 4. Consider the reaction shown below: NH2-(ag) + H2O(l) ê·¼ NH3(gg) + OH-(a...
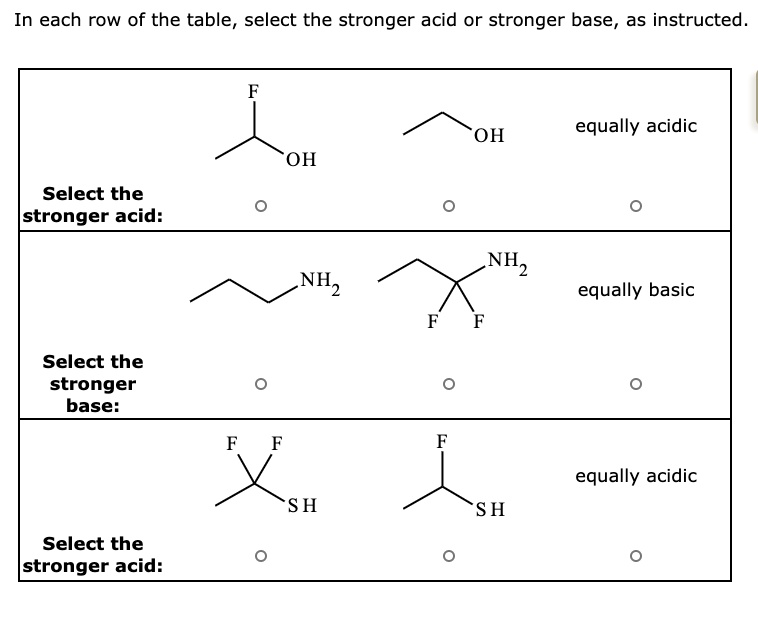
SOLVED: In each row of the table, select the stronger acid or stronger base, as instructed. equally acidic OH OH Select the Istronger acid: NH equally basic Select the stronger base: equally

a) Mention conjugate base of each of the following: HS^-,H3O^+,H2PO4^-,HSO4^-,HF,CH3COOH,C6H5OH,HClO4,NH4^+ (b) Mention the conjugate acid of each of the following: OH^-,CH3COO^-,Cl^-,CO3^2 - ,H2PO4^-,CH3NH2,CH3COOH,NH2^- (c) Which of the following ...

SOLVED: Match each substance in the following equation as Acid (A), Base (B), Conjugate Acid (CA), or Conjugate Base (CB) CH3OH NH2 CH3O" NH3 Conjugate Acid CHzOH Acid NH2 Conjugate Base CH3o-
![CH3 - OH|CH - CH2 - CH2 - NH2 [ Triethylamine ]Ethyl formate Product The major product of the given reaction is: CH3 - OH|CH - CH2 - CH2 - NH2 [ Triethylamine ]Ethyl formate Product The major product of the given reaction is:](https://dwes9vv9u0550.cloudfront.net/images/7494062/22483b28-e987-494e-85b6-898b531ad092.jpg)
CH3 - OH|CH - CH2 - CH2 - NH2 [ Triethylamine ]Ethyl formate Product The major product of the given reaction is: